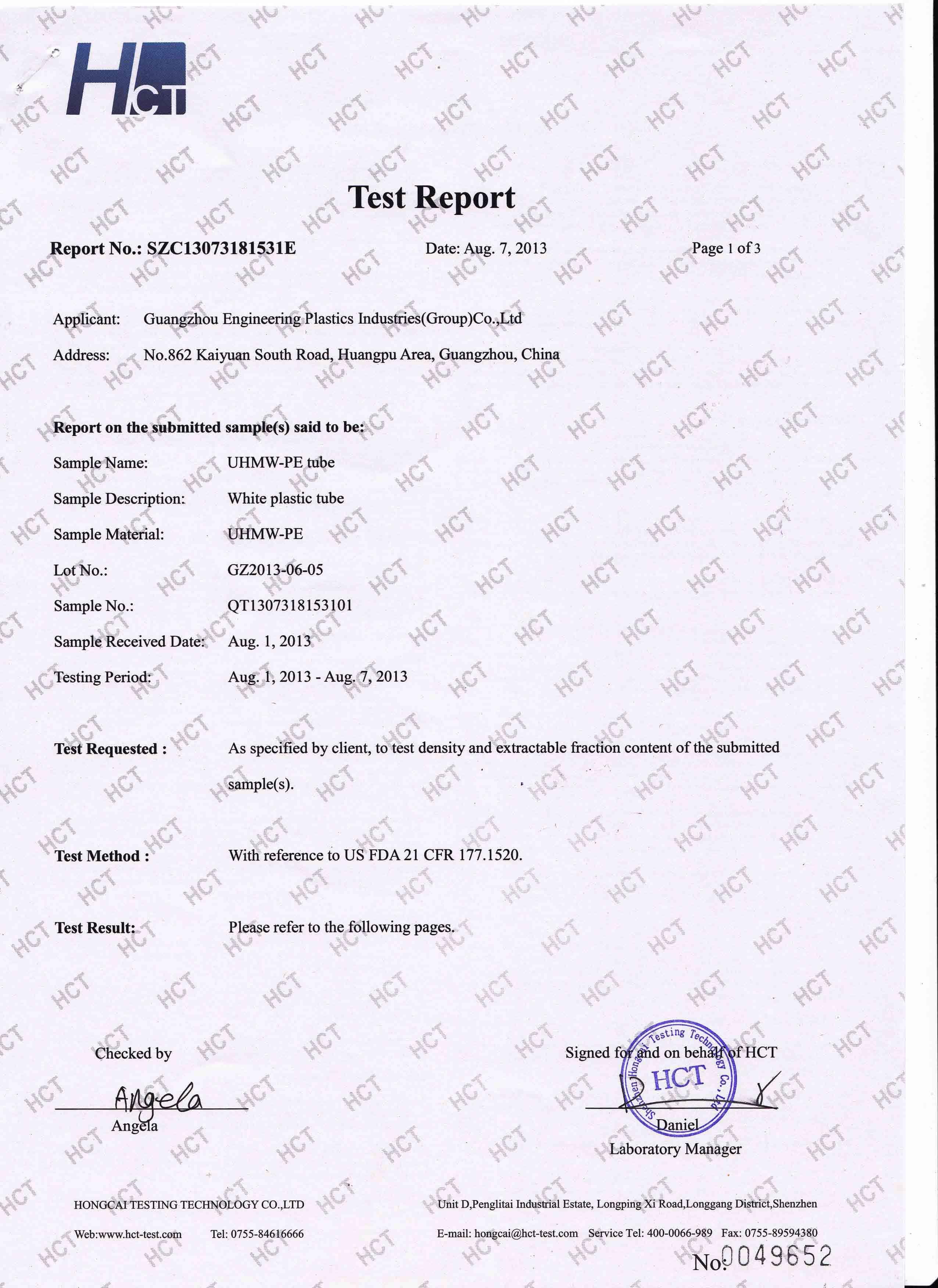
fda certificate
Through the certification of major institutions
Our company has passed ISO9001 quality system standard certification, FDA of the United States, CE certification of the European Union and so on. Our company focuses on engineering plastics, special plastics and other plastic industry fields, high requirements, high standards for your custom plastic parts escort!
The Food and Drug Administration (FDA or USFDA) is a federal agency of the United States Department of Health and Human Services, one of the United
States federal executive departments. The FDA is responsible for protecting and promoting public health through the control and supervision of food safe
ty, tobacco products dietary supplements, prescription and over-the-counter pharmaceutical drugs (medications), vaccines, biopharmaceuticals, blood tra
nsfusions, medical devices, electromagnetic radiation emitting devices(ERED), cosmetics, animal foods & feed and veterinary products. As of 2017, 3/4th
of the FDA budget (approximately $700 million) is funded by the pharmaceutical companies due to the Prescription Drug User Fee Act.
The FDA was empowered by the United States Congress to enforce the Federal Food, Drug, and Cosmetic Act, which serves as the primary focus for the
Agency; the FDA also enforces other laws, notably Section 361 of the Public Health Service Act and associated regulations, many of which are not direc
tly related tofood or drugs. These include regulating lasers, cellular phones, condoms and control of disease on products ranging from certain household
pets to sperm donation for assisted reproduction.
The FDA is led by the Commissioner of Food and Drugs, appointed by the President with the advice and consent of the Senate. The Commissioner reports
to the Secretary of Health and Human Services. Scott Gottlieb, M.D. is the current commissioner, taking over in May 2017.
The FDA has its headquarters in unincorporated White Oak, Maryland.[8] The agency also has 223 field offices and 13 laboratories located throughout the
50 states the United States Virgin Islands, and Puerto Rico] In 2008, the FDA began to post employees to foreign countries, including China, India, Costa
Rica, Chile, Belgiumand the United Kingdom